DEEPBLUE Coronavirus (COVID-19) antigen detection reagent-saliva has passed CE1434 certification, and its comprehensive strength has been internationally recognized
Nov 19, 2021Anhui Deepblue Medical Technology Co., Ltd. (hereinafter referred to as "Deepblue Medical") has made great achievements in expanding the market recently. Its domestic business is booming, and its foreign trade business is constantly breaking through. In order to better serve the EU market, Deepblue Medical has been deeply engaged in the field of technology research and development. After unremitting efforts, the self-developed novel coronavirus (COVID-19) antigen detection reagent-saliva has successfully obtained the CE1434 certificate on November 10, 2021.

Deepblue medical novel coronavirus (COVID-19) antigen detection reagent-saliva successfully obtained the CE1434 certificate.
So far, Deepblue Medical has passed CE1434 certification for 3 types of novel coronavirus (COVID-19) products, mainly including novel coronavirus (COVID-19) neutralizing antibody (double antigen sandwich) detection reagents, novel coronavirus (COVID-19) antigen detection reagents -Anterior nose, novel coronavirus (COVID-19) antigen detection reagents-saliva, so far, Deepblue Medical is one of the only IVD companies in China that have obtained three certificates at the same time. Deepblue Medical has a wealth of novel coronavirus products and detection solutions. Help global epidemic prevention and control.
Deepblue Medical Novel Coronavirus (COVID-19) Antigen Saliva Detection Reagent can quickly detect whether saliva contains nocel coronavirus through saliva samples collected from individuals. It is a very suitable product for personal and family self-testing. It has the following main features Features:
Convenient sampling
Convenient and fast
The results are accurate
Since the early stage of the outbreak of the novel coronavirus, Deepblue Medical has integrated its technical strength, and its self-developed series of novel coronavirus (COVID-19) antigen detection reagents have been verified by multiple authoritative institutions: through the British Phase 3B test, the German BfarM special authorization, the Netherlands MINVWS special authorization, etc. The CE1434 certification of the series of novel coronavirus products not only reflects that the product's excellent performance and good user experience have been fully recognized, and meets the needs of the international market, it is also a favorable evidence for Deepblue Medical's influence in the IVD industry, international customer recognition, and international competitiveness. .
Deepblue Medical Novel Coronavirus (COVID-19) Antigen Detection Reagent - Saliva Detection Reagent
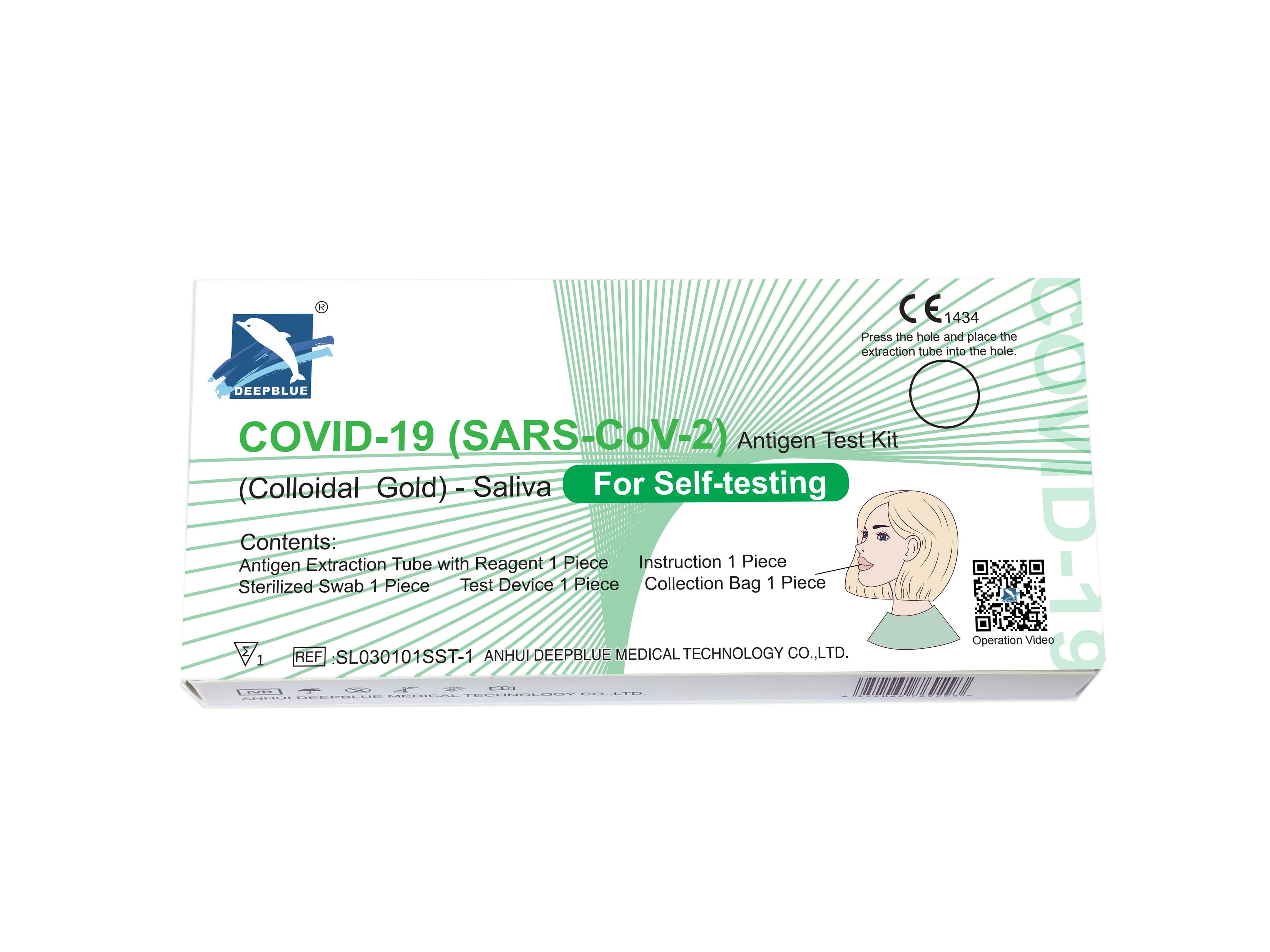
Relying on professional scientific research and innovation strength, Deepblue Medical has independently developed novel coronavirus (COVID-19) antigen saliva detection reagents in a faster and more convenient way to meet the needs of individuals and families for self-testing, allowing more people to cut the infection chain faster , to prevent the risk of virus transmission.
In addition, Deep Blue can also provide a full range of rapid detection solutions covering novel coronavirus antigens, antibodies, neutralizing antibodies, nucleic acid detection, etc. to meet the testing needs in different scenarios. Deepblue Medical adheres to the construction of quality system and certification system. The quality has been recognized by many overseas countries and regions. In the future, Deepblue Medical will continue to make greater contributions to the development of the IVD industry with continuously innovative products and services under the leadership of the development of the health industry.